| 
|  |
| Artists & Craftsmen on the Lunar Frontier | |
![]() The Role of Artists & Craftsmen on the Lunar Frontier
The Role of Artists & Craftsmen on the Lunar Frontier
The rocket scientists may get us there, and even give us a way to hop around globally. But it is the chemical engineers who will figure out how we can "live off the land", giving us building materials, manufacturing stuffs, and craft materials. It is the entrepreneur who will find ways to make a buck for all of us out of these elements. It is the architect who will find a way to turn these prepared stuffs into habitual shelter, and the pressurization engineer who will find a way to keep the biosphere from leaking out. It is the biosphere engineers and agricultural people and waste mass processors who will find ways to keep us alive inside.
![]() But it is the artists and craftsmen especially, who, finding ways to give creative expression to their talents using locally derived materials, who will give us that sense of being home, dealienizing the Moon, because they will have learned how to make moonstuffs over in our own image and likeness.
But it is the artists and craftsmen especially, who, finding ways to give creative expression to their talents using locally derived materials, who will give us that sense of being home, dealienizing the Moon, because they will have learned how to make moonstuffs over in our own image and likeness.
![]() The Challenge for Early Lunan Artists
The Challenge for Early Lunan Artists
How could lunar pioneers express themselves in a paint medium derived entirely from local materials. The assumption [see first * asterisk note below] is that organic materials would be scarce or excessively expensive. Use of substances produced as agricultural byproducts in the settlement biosphere might be frowned upon - the rule being to plow everything back into the food chain and general biosphere support.The rubric chosen by the experimenter was to exclude all organic materials, and include only those inorganic materials that could eventually be processed by a lunar settlement from common regolith soils.
The spark that eventually led to this experiment was our stumbling upon the remark that "sodium silicate is the only known inorganic adhesive." Could "paints" be made using an adhesive as a medium rather than a solvent? All the elements in sodium silicate [sodium, silicon, oxygen] are commonplace in representative lunar regolith soils. If so, common metal oxides producible from the regolith could serve as pigments.
Fortunately, there was a local supplier of chemicals, Laabs Pharmacy in Milwaukee, that was able to help me round up what was needed: a gallon of sodium silicate [$9.95 a gallon, characterized as "pure Na2O-3.75SiO2, no water added"], and small amounts of various metal oxides. We wish to acknowledge Tom Volkman, head chemist at Laabs, who was most cooperative in our project. Yes, we did tell him what it was all for, once we had successfully produced the first painting. - P. Kokh
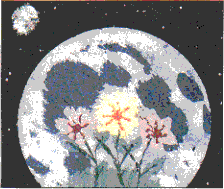
click to enlarge
"Moon Garden #1"
A very unsatisfactory scan.
There are no bluetones in the 8"x10" originalPossibly the first painting done exclusively with materials that could be processed from the regolith soils in an early lunar settlement. Moon Garden #1 shows a plot of grass (light green) with three flowers (light green leaves, dark green stems; mid: orange center with yellow petals; r & l: rust center with pinkish petals) superimposed on a full Moon against starry sky with full Earth in corner.
Backpainted on glass, the paints consist of ferric and chromium oxides, manganese and titanium dioxides, and sulfur in a medium of sodium silicate.
by Peter Kokh, September 30, 1994
- 1630 N. 32nd St.,
- Milwaukee WI 53208-2040
- (414) 342-0705
Metal oxides in a waterglass (sodium silicate) suspension, backpainted on glass, foreground first.
This painting is the first ever attempted, to my knowledge by anyone, using exclusively art materials/media that could be produced in an early lunar settlement, processing the local regolith dust. My purpose is to pioneer a medium future lunar settlers can use.
Sodium Silicate, a cousin of window glass, and, when hydrated, a liquid at room temperatures (keep bottle tightly closed when not in use, lest it begin crystallizing) is the only known inorganic adhesive. The water of hydration is the only available solvent. This is the basis of what I call waterglass "paints" or waterglazing. To this is added available colored powders.
"Greening the Gray", our second piece
![]() A Pioneer Palette of "Moontones"
A Pioneer Palette of "Moontones"
- The light to very dark gray lunar regolith powder soils are, of course, already available as is. While I do have a pinch of lunar simulant, my grays are mixes of
 (black) and
(black) and  white.
white.  gives a rust, and when added cautiously to a preparation of white, produces the pinkish to rust colors in the painting.
gives a rust, and when added cautiously to a preparation of white, produces the pinkish to rust colors in the painting.- The pale yellow is
 . With cautious amounts of iron oxide mixed in, orange hues are produced.
. With cautious amounts of iron oxide mixed in, orange hues are produced. - For green, I used
 as is for veining, and pasteled with titanium dioxide and/or sulfur for leaves and grass.
as is for veining, and pasteled with titanium dioxide and/or sulfur for leaves and grass. - At first I found no lunar-producible blue. There seemed to be a couple theoretical possibilities such as cobalt aluminate, but I had not yet found a source. Accordingly, I decided to use a turquoise for the Earth in the background, upper left, made with nickel sulfate (copper is present on the Moon only in parts per billion traces, so far as we know). Unfortunately, the the nickel sulfate reacted chemically with both the waterglass and the neighboring oxides, messing up my "Earth".
- Eventually, I found an extremely pricey ($121/28 grams)
 that provided a stunning bright blue.]
that provided a stunning bright blue.]  Some powders mix readily, producing a workable paste for painting. Others tend to coagulate almost immediately and applying them is more a matter of dabbing chunks of it to the glass before the sodium silicate adhesive dries. Three of the powders I have experimented with to date are in this difficult category: nickel sulfate, potassium chromate (bright yellow) and chromium trioxide (magenta).
Some powders mix readily, producing a workable paste for painting. Others tend to coagulate almost immediately and applying them is more a matter of dabbing chunks of it to the glass before the sodium silicate adhesive dries. Three of the powders I have experimented with to date are in this difficult category: nickel sulfate, potassium chromate (bright yellow) and chromium trioxide (magenta).For a canvas, settlers can use glass (front side, or back as here), unglazed ceramic tile, or metal sheet. I hope to continue experimenting.
Again, this is the very first known complete work with "paints" and "canvas" made of materials locally producible in a lunar settlement.
![]() The idea behind this project is rooted in two premises:
The idea behind this project is rooted in two premises:
- (1) lunar settlers will have to keep imports to the bare minimum of absolute essentials, and try to make do with what they can make from local materials.
- (2) their garden / farm / biosphere, as it depends on carbon*, nitrogen*, and probably hydrogen* from Earth [as well as local lunar oxygen and many nutrients] will be designed to produce only food and clothing fibers, everything being tightly recycled. It won't be permitted to withdraw art materials and ingredients from the cycle.
NOTE: Lunar Prospector's confirmed discovery in 1998 of ample comet-derived water ices (probably with immixed carbon oxide and nitrogen ices) at both Lunar poles in permanently shaded crater areas may, in time, greatly relieve this second constraint.]
An article ("The First Lunan Artist") about this painting and the effort behind it, was published in Ad Astra, tin the January-February '95 issue, pp. 46-47. A color photo of the painting was included. Ad Astra is the magazine of the National Space Society.]
 large image of cover no longer available
large image of cover no longer available
![]() "Regolith Impressionism"
"Regolith Impressionism"
Sodium Silicate is not easy to work with. You have to mix small less than teaspoonful amounts of the adhesive and metal pigment powder at a time and use it quickly, as it sets up fairly fast. I had to plan the painting carefully, so that, if practical, everything to be painted in a given color was done at the same time. Because the medium is so viscous, fine controlled detail is not possible. The painting that results will have much fine detail, but it will be largely serendipitous. Painting on the reverse side of the glass pane, as I had chosen to do, meant having to plan carefully - items in the foreground of the picture had to painted first, those in the background last. If there were void spot or streak "skips" in the "paint" as it dried on the glass (in moments) a second cover coat in another color or highlighting shade would produce welcome veining and texture visible from the front. The effect is somewhat impressionistic and indeed, slavish realism and accuracy are out of the question. As the pigments and medium and glass 'canvas' are all producible from the elements common in the pulverized powdery regolith soils of the Moon, I came to call my painting (at first dubbed "waterglazing") as "Regolith Impressionism", a fitting description.The idea of gallons of ready-to-use sodium silicate metal oxide paints being produced for the purpose of painting whole walls, for example, is out of the question. Painting an 8"x10" pane or smaller is just about right. One could, of course, paint a whole wall, if one wanted to take the time and exercise the patience and determination. Quilt-like murals might be just the ticket. Even "sponge painted" wallpaper like designs would be difficult. The flaking problem described below would have to be fixed before it would be worth making an attempt.
![]() L.A.A.M.P. - Lunar Appropriate Art Media Pioneers
L.A.A.M.P. - Lunar Appropriate Art Media Pioneers
It was desirable to get others involved, hopefully others with more practiced and highly developed artistic painting skills than mine - to see to what heights this new pioneer-appropriate medium might be pushed. The driving spark was twofold. (1) create, field test, and pre-debug a viable art medium for early lunar pioneers, and (2) capture the public imagination by demonstrating one small aspect of the coming lunar frontier in vivid concrete terms. To do this, we launched a special newsletter, Moonbow, for those who donated to our fund to buy more experiment chemicals. But there were few takers, and only two issues were ever circulated.
![]() Decay of the Paintings over Time
Decay of the Paintings over Time
We produced a number of additional paintings over the following year (late '94 and throughout '95). But then we were disturbed to notice that after 6-10 months, a progressive deterioration set in. The "paint" began to "flake off" the glass 'canvas'. (I had also tried painting on terra cotta flower pots, aluminum, and brick, with similar results - in time the paint would crystallize and rub off. Was sodium silicate a temporary adhesive after all? If so, it could still be used by Lunan artists, but only for temporary art du jour. Were there additives unknown to me that would fix the problem? I stubbornly resisted resorting to using anything organic in nature as a deus ex machina. True, lunar pioneers might be able to afford minor amounts of some rescue organic substance imported from Earth. But I resisted giving in to that approach, wanting to continue to see how far this experiment could be pushed, being faithful to the "no-organics" guidelines I had adopted.Not being more than a very good high school level chemist, I had no clue to what ailment afflicted my paintings. I eventually abandoned the effort. Now [early 1999], however, I am ready to try again. I will attempt "Moon Garden #2" (incorporating the cobalt blue, this time!), but adding a second pane as a backing as soon as the paint had dried thoroughly on the first (a half hour), and then sealing the gap between the two panes along the edges with a semi-organic silicone adhesive. Then we'll wait a year and see what happens. - PK
![]() In early 2000, Ron Zdroik, a commercial artist who is a fellow member the Wisconsin Mars Society, suggested that I should try micro-etching the glass with wet metal oxide paper first, and that my delamination problem might go away or be abated.
In early 2000, Ron Zdroik, a commercial artist who is a fellow member the Wisconsin Mars Society, suggested that I should try micro-etching the glass with wet metal oxide paper first, and that my delamination problem might go away or be abated.
"Red Sands, Blue-Green Dreams"
On June 14th, 2000, we produced the above new painting, and sent it along with a set of Earth-Mars-Moon "Gravity Bricks" requested by Dr. Pascal Lee, to be taken up to Haughton Crater on Devon Island for the openning of the Flashline Mars Arctic Research Station. In this painting, a "regolith impressionist" rendering of a marsscape of today, is matted with a continuation of the scene in the colors of a Mars that has been terraformed.
UPDATE
![]() Megan Storrar of Toronto brings a fresh hand to the experiment, producing two beautiful pieces. - July 27, 2000
Megan Storrar of Toronto brings a fresh hand to the experiment, producing two beautiful pieces. - July 27, 2000
|
| |
|
Her work demonstrates that this "crude" frontier medium is capable of much finer detail and refinement than we had thought.This is a superb example of why it is important to involve other, more accomplished artists in this experimentation.
If you would like to try your hand and skill, and artistic instincts to this pioneer medium, please contact us.
![]() More Orphaned Lunan Arts & Crafts Homework
More Orphaned Lunan Arts & Crafts Homework
There are other potential media for early lunar pioneers that those with the talent, curiosity, and determination to help, can investigate. But many of them will involve greater expense than the $300 some spent on the project above. Making art glass out of simulated lunar materials, for example. Seeing what can be done with cast basalt is another. If you have a special expertise in some craft media that might possibly have an application on the Moon and are willing to pursue this avenue further, contact us for some ideas.A list of Subject-Related Articles in Moon Miners' Manifesto is given at the bottom of this linked page.
![]() Previous Work with "Waterglass / Metal Oxide Paints"
Previous Work with "Waterglass / Metal Oxide Paints"
Gerald J. Grott writes (Sept. 16, 2001) the following:FYI-----The first recorded origin of painting with waterglass and inorganic pigments was about 1840. It was known as 'stereochromie' and most university libraries have one or more references under that name.Hundreds of buildings in middle Europe still sport external paintings in bright colors though they are over a century old.
The shroud of Turin, scores of feet high, was painted on linen and hung in a German Cathedral until destroyed by Allied bombs in WWII.
I myself started with this about 50 years ago. My original purpose was to flame proof wood with bright colored paint that soaked in.. It worked very well as, on exposure to high heat, the wood would char but not burn.
In the 1970's we started a new business to commercialize the matching of the natural colors of rocks, particularily "Desert Patina", so that rock surfaces exposed by earth moving and blasting can be economically restored to a permanent matching surface coloration. We purchased the sodium silicate in numbers of 55 gallon drums.
Unfortunately, our young manager died of cancer and none of us chose to leave our own businesses to run that one and we let the business die.
For painting pictures and illustrations, most any of the truly insoluble inorganic pigments are compatable with sodium silicate. However,you must be very careful not to have any contamination with soluble carbonates or sulfates. These are in detergents and soaps so you must rinse surfaces carefully before painting.
Also, avoid painting on cloths that have sizing in the fabric. Sizing in new cloth will almost always cause flaking or other decrepitation of the silicate.
As history shows, unsized linen is a good base.
Magnesium oxide is a good material for reacting slowly with sodium silicate to form a 'permanent' rock like coating.
I have several full sixed notebooks of R&D regarding the use of sodium silicate base nmaterials for sealing surfaces against moisture penetration and particularily for avoiding deterioration of marble objects or masonry of, or containing limestone or magnesite.
You are on a good course. I wish you good luck.
Jerry Grott
P.S. The "bible" in this matter is a book called Soluble Silicates and any good University Library will have it if they have a Chemistry Dept. It has the history of stereochromie in it. It also has a lot of info about stabilizing silicates.
Also try Philadelphia Quartz Technical Services for info about misadventures with sodium silicate. They are not usually heavy on art but they are highly knowledgeable about what affects the performance of their silicates. - JG
![]() LINKS to Relevant Pages and Sites - U.C. (under construction)
LINKS to Relevant Pages and Sites - U.C. (under construction)
Lunar Reclamation Societ Pages below
Lunar Painting Experiment
Peter Kokh ![]()